Galderma’s newest hyaluronic acid lip filler, Restylane Kysse, is more than meets the eye. We sat down with Alisa Lask, general manager and vice president of the U.S. Aesthetic Business at Galderma, to find out exactly what the latest filler on the market has to offer. Spoiler alert: it’s a lot.
A smaller amount of filler is required
Findings from Galderma’s multi-phase study were surprising—the filler was found to last up to a year, for one—but the most shocking finding, according to Lask, didn’t have to do with longevity. “The results that surprised us most came from the Phase 3 head-to-head study that showed a significantly lower amount of Restylane Kysse was needed to show an improvement in lip fullness when compared to the control filler (1.82mL vs. 2.24mL),” she explains.
The results are a crowd-pleaser
Lask says another interesting finding came from the brand’s Phase 4 study, which revealed that 90-percent of patients’ partners were satisfied with the results of Restylane Kysse treatment. “We know from market research that patients can be reluctant to get lip injections because they are concerned with partner satisfaction, so we are thrilled with this finding.”
Kysse is the first FDA-approved lip filler to use XpresHAn Technology
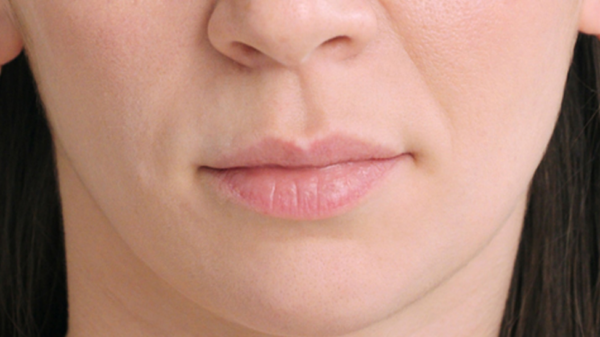
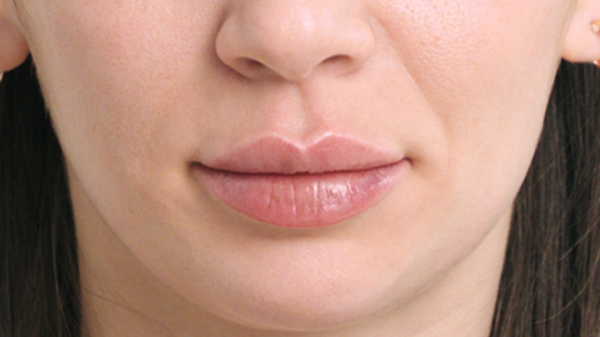
While Lask explains Restylane Refyne and Defyne, both approved for laugh lines, are formulated with XpresHAn Technology—a manufacturing process which Lask says allows for a gel that “integrates into the skin for natural expression in motion” versus a firmer, more structured look—Restylane Kysse is the first and only FDA-approved lip filler to harness the technology. The result? A plush, pillowy look, as seen in the below brand-supplied photos.
From: https://www.newbeauty.com/fda-approves-restylane-kysse/